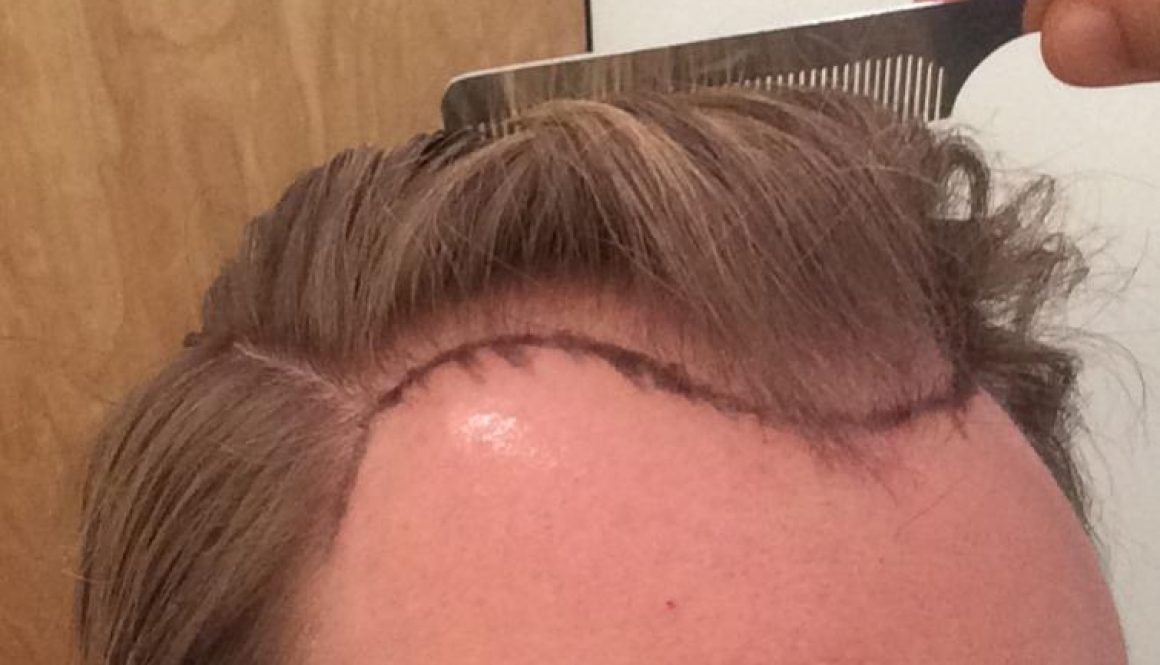
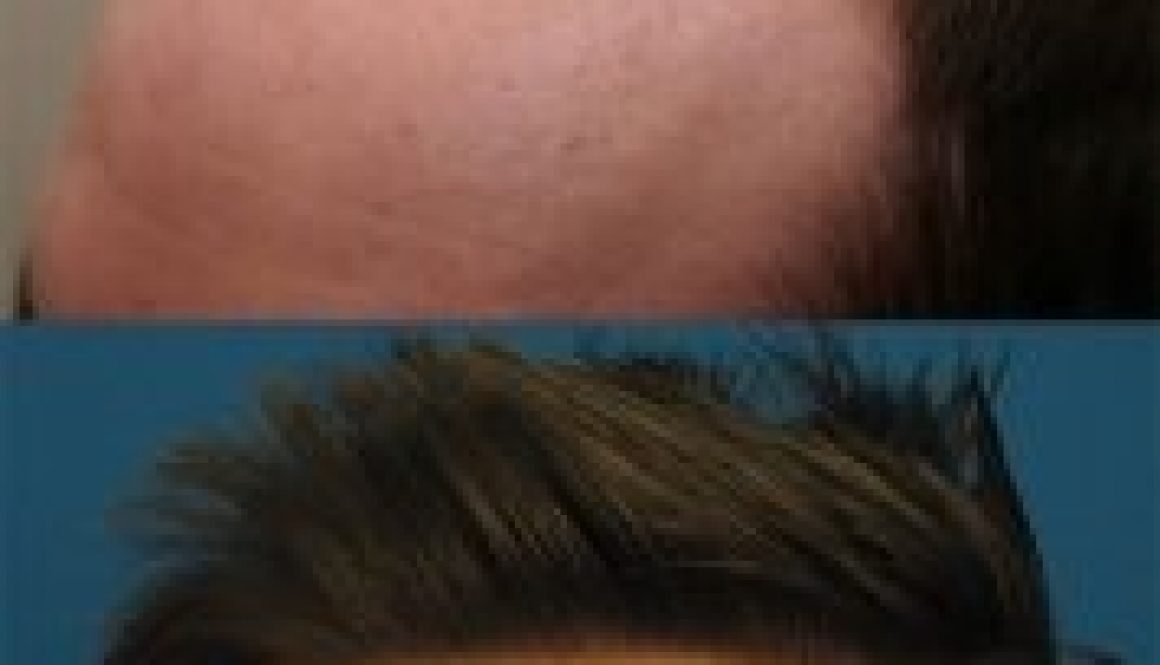
Body Hair Transplants Are They Worth It? Dr. Pradeep Sethi Answers

I’m at thirty-one year old man with a Norwood 6 level of hair loss. I have been losing hair since I was in my late teens. I’m now interested in getting a hair transplant, but many clinics have turned me away. I have now researched body hair transplants (BHT), but I don’t know if it’s […]